Mesoamericana, 27 (1): marzo-agosto 2025 pp.35-41
ARTÍCULO CIENTÍFICO
SALVEMOS AL CARACOL REINA, ALIGER GIGAS (LINNAEUS, 1758) EN NUESTRAS COSTAS PANAMEÑAS
Universidad de Panamá, Facultad de Ciencias Naturales, Exactas y Tecnología, Panamá.
dario.cordoba@up.ac.pa https://orcid.org/0000-0002-9460-1717
DOI https://doi.org/10.48204/j.mesoamericana.v27n1.a7368
INFORMACIÓN SOBRE EL ARTÍCULO
Recibido: 3 de enero 2025 | Aceptado: 24
febrero 2025 | Publicado: 30-marzo 2025.
Como citar este documento: Córdoba González, Darío. 2025. Let's save the queen conch, Aliger gigas (Linnaeus, 1758) on our Panamanian coasts. Mesoamericana 27(1): X-X.
Autor corresponsal: Darío Córdoba González, Universidad de Panamá, Facultad de Ciencias Naturales, Exactas y Tecnología, Escuela de Biología, Departamento de Zoología, Museo de Malacología de la Universidad de Panamá (MUMAUP), Miembro del Sistema Nacional de investigación SIN-SENACYT, Panamá., dario.cordoba@up.ac.pa
Contribución del autor: El autor de este trabajo declaran haber participado en la realización de este proyecto de investigación en todas sus etapas, búsqueda de información y redacción del artículo.
Editor: Alonso Santos Murgas.
ABSTRACT: A summary of the state of knowledge of the biology and fisheries of the gastropod mollusk Aliger gigas (queen snail, Caribbean conch) in the Panamanian Caribbean is presented, and the use of the resource by artisanal and industrial fishing is highlighted. The biological and population status detected through evaluations carried out in both insular and continental areas is also analyzed, and finally, recommendations are made on some management and planning measures that should be immediately implemented, because if they are not implemented, the resource that is threatened will enter a process of extinction in the short term.
KEYWORDS: Aliger, snail, mollusk, shell, Caribbean.
RESUMEN: Se presenta una síntesis del estado de conocimiento de la biología y la pesquería en el Caribe panameño del molusco gasterópodo Aliger gigas (caracol reina, cambombia del caribe), y se resalta el aprovechamiento que se hace del recurso por parte de la pesca artesanal e industrial. También se analiza el estado biológico y poblacional detectado a través de las evaluaciones realizadas tanto en las áreas insulares como continentales y, finalmente, se hacen recomendaciones sobre algunas medidas de manejo y ordenamiento que deben ser de inmediata implementación, pues de no hacerlo, el recurso que está amenazado entrará en proceso de extinción en el corto tiempo.
PALABRAS CLAVE: Aliger, caracol, molusco, concha, Caribe.
The genus Strombus Linnaeus, 1758 includes species widely distributed in the shallow waters of the Caribbean and the Tropical Pacific, where it is found from Baja California to Peru. Some of the species have enormous commercial importance (Keen, 1971), fisheries in the Bahamas and Caicos Islands allow the export of more than 3.9 million individuals annually (Doran, 1958; Iverson, 1968; Boss 1969). In the Bahamas, its fishery occupies second place and in the American Virgin Islands, third place as the main fishing product (Damman, 1969).
The Strombus gigas (Linnaeus, 1758) (Gastropoda: Caenogastropoda: Strombidae) previously belonged to these genera, but has now been changed to the genera Aliger Thiele, 1929; it is used as food in all its sizes, according to its abundance and local preferences (Randall 1964). The bright color of the pearly layer, its size and the quality of its meat are what have stimulated its collection and consumption, which has resulted in overexploitation of many populations, particularly those in the Caribbean and the Mesoamerican Pacific (Villalobos, 1986).
Although A. gigas has been studied since the 1960s, and more than 600 scientific articles have been reported to date (Darcy, 1981; Acosta, 1994; Ray-Culp, 2002), there is a lack of knowledge about its basic histology.
A. gigas is a gastropod mollusk of the Strombidae family. It is distributed in the southern part of the Florida peninsula, the Bermuda and Bahamas islands, the Caribbean coasts of Mexico, Central and South America and the entire Antilles arc (Randall,1964, Brownell and Stevely, 1981).
Adult snails reproduce all year round, with greater reproductive activity between spring and autumn. On average, a female produces about 500,000 eggs in each ovigerous mass and can spawn repeatedly throughout a year, so its annual production of larvae reaches several million (Randall, 1964). Individuals emerge from the
egg as planktonic larvae (“veliger”). The larval stage of
A. gigas lasts about 3 to 5 weeks, during which time they are transported by ocean currents (Ballantine and Appeldoorn, 1983), so it is presumed that their mortality rates are very high. The last larval stage undergoes a metamorphosis towards the juvenile stage with benthic habits. The smallest juveniles have cryptic habits and spend much of their time buried in the sand (Iversen et al., 1990). As they grow, the edge of the shell opening of A. gigas snails grows and extends, forming what is known as a “fan”, and later the lip or edge of said fan thickens (Randall, 1964). Although the age of sexual maturation is around 3 to 4 years (Berg and Olsen, 1989), it has been determined, through the analysis of the presence of secondary sexual organs and the histological analysis of the gonads, that only those individuals with a lip that is 4 mm or thicker are sexually mature (Berg, 1976; Berg and Olsen, 1989), and that once sexual maturity occurs, the shell stops growing in length. Therefore, age and sexual maturity are not related to shell length, so it is common to find mature individuals (with a thick lip) and immature individuals (with a thin lip) overlapping in a wide range of shell lengths in A. gigas populations. Due to this situation, frequency analyses based only on shell length do not provide a true representation of the composition of an A. gigas population. In addition, snails. gigas show a great variability in the length and shape (sharp or blunt) of the spines, and in the density of the shell (Alcolado, 1976; Martin-Mora et al., 1995).
Queen conch feed on algae and organic detritus. Algae may be small interstitial algae that proliferate between sediment grains or epiphytes that grow on seagrass leaves. Therefore, these snails are most found in sandy areas and seagrass beds (Stoner and Waite 1990).
Smaller juveniles have numerous predators, such as brachyuran crabs (families Calappidae, Portunidae, Majidae), various species of hermit crabs; caridean shrimps (Alpheidae and Palaemonidae), lobsters (Palinuridae), polychaeta worms and fish (Ray-Culp et al., 1997). Larger animals within some of these taxa
(such as Panulirus argus) may also prey on larger juveniles (Delgado et al., 2002). However, the vulnerability of A. gigas to its predators decreases as its size increases (Jory and Iversen, 1983).
There are different species of Strombus in the Republic of Panama; Titanostrombus galeatus (Swainson, 1823) and Lobatus peruvianus (Swainson, 1823) are found in the Pacific sector, and in the Caribbean species such as
A. gigas, Lobatus raninus (Gmelin, 1791) and Macrostrombus costatus (Gmelin, 1791) have been identified.
In the Atlantic zone, regions have been located where marine snails of the species A. gigas are collected all year round; in the Province of Bocas del Toro, close to the protected areas of the Archipelago; specifically on Escudo de Veraguas Island, Cayo de Agua, Bahía Azul and Punta Valiente; and in the region of San Blas.
Since fishing for this important marine product is not regulated by law, Executive Decree No. 159 of December 31, 2003, established a ban on this snail species in the jurisdictional waters of the Republic of Panama for a period of 5 years. However, there are indications that certain illegal fishing activities are still taking place for local consumption in the areas described above.
Years ago, this species was found at a depth of 3 meters, however, some fishermen have stated that lately this species is found at depths greater than 20 meters, because of continuous fishing.
The extraction of cambute or queen snail is generally carried out by lung, without a diving tank, since it is prohibited; generally this activity is carried out by indigenous artisanal fishermen from the Guna Yala region, who are excellent divers, since their capacity for resistance is greater compared to local divers; the extraction is carried out by ancestral tradition; They move in groups of 20 to 40 divers.
The “cambombia”, the common name given to the Queen Snail in Panama, is usually consumed in places visited by tourists who see this resource as an
aphrodisiac. It is generally presented in delicious dishes in hotel restaurants. Although it has not been possible to compile information on the volume of local consumption, there is an irregular annual record, by ton from 1984 to 1991. Regarding local sales, divers sell the snail directly by the pound to middlemen, hotels and restaurants; the estimated price on land is B/. 1.50 - 2.00 per pound (weight of the meat with viscera).
The populations of A. gigas are overexploited, worldwide, regionally and in Panama, and it is necessary to exhaust all measures to protect the species. That is why the purpose of this manuscript is to try to raise the alarm in our authorities such as MiAmbiente and the Aquatic Resources Authority of Panama (ARAP) to create a mechanism to protect the species so that it does not fall into any CITES category.
Between March 1995 and December 1997, several snail samplings were carried out to collect morphometric data and tag individuals, as well as some complete visual censuses. The samplings were carried out by means of SCUBA diving. For comparative purposes, samples were taken separately on the coasts of two provinces and one region of the Panamanian Caribbean (Comarca Guna Yala, Colón and Bocas del Toro).
Sampling in the Marine Zone (MZ)
In the MZ, samples of A. gigas were taken both on sandy bottoms and in seagrass beds, within the area between the rocky coast and the buoys that mark the marine limit of the mentioned zones (i.e., up to a depth of 7 to 10 m). Eight samplings were carried out at irregular intervals in this environment between April 1996 and December 1997. Four dimensions of the shell were measured for each snail (Lara-Pérez Soto, 1992; Tewik and Guzmán, 2003). The total shell length (TSL), measured between the apex and the posterior end of the shell, was obtained with an ichthyometer (± 5 mm), while a vernier caliper (± 0.1 mm) was used to measure: the lip thickness (LT), at the height of the middle part of the shell fan; the cone width (AnCo), at the widest part of the shell cone, excluding the aperture and the fan; and the shell aperture
(AbC) measured at its most open part; to determine whether they were juveniles (immature) or adults (mature) (Fig. 1). All dimensions are expressed in millimeters. To analyze the data, the snails were classified into two ontogenetic, considering as adults all those snails with a lip thickness ≥ 4 mm and as juveniles those individuals smaller than the minimum LTC of adults.
Figure 1.
a. queen snail or “Cambute”, Aliger gigas Linnaeus, 1758 in Thalasia meadows; b. immature and mature phases. Photo courtesy of Richard S. Appeldoorn
 |
 |
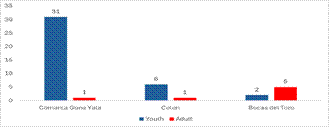
A total of 46 specimens of A. gigas were collected in the three Caribbean sites, of which 41 were juveniles and seven were adults; the Guna Yala region was where there were more juveniles and the Escudo de Veraguas
Island, Bocas del Toro where there were more adults (Fig. 2).
Figure 2.
Number of juvenile and adult specimens of
A. gigas in the different sites of the Caribbean of Panama.
 |
Populations of Strombus spp. have been recorded in 31 countries in the Southwestern Caribbean, which are South and Central America, Florida and the Lesser Antilles (CITES, 1995). Currently, the distribution of A. gigas in the Caribbean is from Florida to the northern coast of South America. It is found in clear waters up to depths of 120 m and sandy seabeds; the reproductive process occurs in the warmer months of the year, although in some areas it can last all year round; also depending on the season its populations can migrate (Appeldoorn, 1993).
It is believed that the natural populations in Bermuda, the Dominican Republic, Florida (USA), Haiti, Costa Rica, Puerto Rico, Trinidad & Tobago, Venezuela and those on the northern coast of Cuba and in the Gulf of Morrosquillo in Colombia have disappeared; although sufficient research has not been carried out to determine the abundance of the A. gigas population; However, in some places in the Caribbean such as the Turks & Caicos Islands, Saint Lucia, southern Cuba and Martinique, there are still populations in good condition (CITES, 1995).
According to Álvarez-León et al. (2007), the species has reproductive strategies that allow it to recover, regardless of the overexploitation, especially of juveniles, which limits the mating capacity of the
specimens, which reduces spawning and the recruitment process. This is why management and planning measures are needed throughout the Caribbean region, to manage the concept of population and not that of the species. For this, its high fertility and its larval cycle of 20 days are being considered. In addition, the species is not imminently threatened, even though demand forces excessive capture.
For many years, A. gigas has been one of the most important fishing resources in the Caribbean, but it has been exploited on a large scale. Currently, it is exploited at around 4,000 tons/year, yielding a commercial profit of around 40 million dollars (CITES, 1996). Because international trade is geared towards meeting the demand for meat, queen conch is being accessed in many islands despite being at depths, a factor along with the alteration of natural habitats that is threatening the stability of queen conch populations (Álvarez-León et al., 2007).
With the exception of Puerto Rico, many countries have legislation for the protection of the species and regulations regarding harvesting; Venezuela, the Virgin Islands, Honduras, Jamaica, Belize, Mexico and Colombia, among others, have a ban; in the Bahamas, the export of meat and shells is prohibited, as is the case in Bermuda; in Belize, the Turks and Caicos Islands, the Dutch Antilles and Colombia, a minimum catch size has been imposed, and in Panama, a five-year ban was established starting in 2020.
Although we see that in the results in the Comarca Guna Yala almost 99% of the individuals collected and measured were juveniles, this is possibly due to the high consumption of the species in this area, it could also be due to the availability of food, since in this place there is a shortage of algae that serve as food for the species; which does not occur on the island of Escudo de Veraguas in Bocas del Toro, where there is more presence of algae that help feed the A. gigas because the human population density in this area is much lower than the other collection sites (Guna Yala Region and Colon) allowing it to reach adulthood. Another aspect to highlight is why more juveniles were found in the
different areas of the Caribbean of Panama; it is because the distribution pattern observed is based on the migratory behavior of the species; because according to Lagos et al. (1996) individuals as they increase in size and mature move towards deeper waters, and we only collect in shallow waters. This migration behavior favors the protection of adult individuals and disadvantages juveniles, since by being in shallow waters they are more accessible to divers, thus contributing to the deterioration of the resource in the long term, since juvenile specimens would hardly manage to reach their reproductive maturity due to overexploitation (Vega and Perez, 2004).
Juveniles of A. gigas consume detritus from seagrass and algae (Randall, 1964; Stoner and Waite, 1991) and as they mature, they move into deeper waters (Weil and Laughlin, 1984; Stoner and Schwarte, 1994). After 3.5-
4 years, the queen or queen conch reaches sexual maturity, its shell stops growing in length and forms the characteristic thickened flared lip of the shell (Egan 1985, Appeldoorn, 1988); its average longevity is at least 6-7 years (Berg 1976), but in deep water habitats it can reach 26 years (Coulston et al., 1987).
Finally, failure to consider management and protection measures for populations, and not just for the species, has led to false economic concepts, which put pressure on and accelerate increased fishing, both artisanal and industrial.
The queen conch, A. gigas, has been subjected to intense exploitation by artisanal fishermen and divers throughout the Caribbean region, including Panama; population densities of this species have decreased and sometimes it has disappeared in coastal areas where it was usually observed. This situation is largely due to overexploitation due to high commercial demand. Its estimated growth rate is between 30 and 90 mm per year, depending on age and environmental conditions, which is equivalent to a monthly growth of between 2.5 and 7.5
mm. Its distribution pattern makes it an organism susceptible to extinction, since it is easier to capture
juvenile and sub-adult individuals, without having reproduced because they are found in shallow or shallow waters, thus reducing its population. Therefore, it is necessary to create protection measures for all existing populations in the Caribbean region where it is found, considering the sizes that can be allowed to be collected.
To all the staff of the Autoridad de los Recursos Acuáticos de Panamá (ARAP) at the laboratory located in Vacamonte for all the support provided in information and logistics during field work, and to the staff of the Malacology Museum of the University of Panama (MUMAUP) for providing all the information for their specialized library.
Acosta A. (1994). Bibliography of the conch genus
Strombus (Gastropoda: Strombidae). pp. 321-
356. En: Appeldoorn, R. S. y Rodríguez, B. (eds).
Proceedings of the workshop on biology,
fisheries, mariculture and management of the
queen conch, Strombus gigas. Fundación
Científica Los Roques, Caracas, Venezuela.
Alcolado P. (1976). Crecimiento, variaciones
morfológicas de la concha y algunos datos
biológicos del “cobo” Strombus gigas L.
(Mollusca, Mesogastropoda). Anales de la
Academia de Ciencias de Cuba, 34: 1–36.
Álvarez-León R., F. Gutiérrez-Bonilla, J.F. Ospina-
Arango and E. Chiquillo-Espítia. 2007. El caracol
de pala (Strombus gigas Linnaeus, 1758) en el
caribe colombiano: revisión monográfica. Boletín
Científico - Centro de Museos - Museo de
Historia Natural, 11: 301 – 332.
Appeldoorn R. S. (1988). Age determination, growth,
mortality and age of first reproduction in adult
queen conch, Strombus gigas L., off Puerto Rico.
Fisheries Research, 6:363-378.
Appeldoorn R. S. (1993). Reproduction,
spawning potential ratio and larval abundance of
queen conch of La Parguera, Puerto Rico.
Caribbean Fishery
Management Council, 25.
Ballantine D. L. and R. S. Appeldoorn. (1983). Queen
conch culture and prospects in Puerto Rico.
Proceedings of the Gulf and Caribbean
Fisheries Institute, 35: 57– 63.
Berg C. J. JR. (1976). Growth of the queen conch
Strombus gigas, with a discussion on the
practicality of its mariculture. Marine Biology,
34: 191–199.
Berg, C. J., JR. and D. A. Olsen. (1989). Conservation
and management of queen conch (Strombus
gigas) fisheries in the Caribbean. Pp. 421–454
En: J. F. Caddy (ed.) Marine invertebrate
Fisheries: their Assessment and Management.
John Wiley y Sons, Nueva York.
Boos K. J. (1969). Conchs. In the Encyclopedia of
Marine Resources. EFL Firth, New York.
p,135-140.
Brownell W. N. and J. M. Stevely. (1981). The biology,
fisheries, and management of the queen conch,
Strombus gigas. Marine Fisheries Review, 43:
1–12.
CITES. (1995). Convención Sobre el Comercio
Internacional de Especies Amenazadas de
Fauna y Flora Silvestres. Aplicación de la
Resolución Con. 8.9. Geneve (Suiza), 30 p.
CITES. (1996). Draft report to the CITES Animals
Committee. Geneve (Suiza), 20 p.
Coulston M. L., R. W. Berry, A. C. Dempsey and P.
Odum. (1987). Assessment of the queen conch
(Strombus gigas) population and predation studies of hatchery-reared juveniles in Salt River Canyon, St. Croix, V.I. Proceedings of the Gulf and Caribbean Fisheries Institute, 38:294-306.
Dammam A. E. (1969). Study of the fisheries potential
of the Virgin Islands. Spec. Rep. Carib. Res.
Inst. 1:1- 204.
Darcy G.H. (1981). Annotated bibliography of the conch
genus Strombus (Gastropoda, Strombidae) in
the Westem Atlantic Ocean. NOAA. Tech.
Rept. NMFS SSRF 748. 16 pp.
Delgado G. A., R. A. Glazer and N. J. Stewart. (2002).
Predator-induced behavioral and morphological
plasticity in the tropical marine gastropod
Strombus gigas. Biology Bulletin, 203: 112–120.
Doran E. (1958). The Caicos trade. Ocoee Rev. 48:388-
401.
Egan B. D. (1985). Aspects of the reproductive biology
of Strombus gigas. Magister thesis, University
British Columbia, Vancouver, B.C., Canada, 147
pp.
Iversen E. S., S. P. Bannerot and D. E. Jory. (1990).
Evidence of survival value related to burying
behavior in queen conch Strombus gigas. Fishery bulletin, 88: 299–309.
Iverson B. S. (1968). Forming the edge of the sea.
Sishing News, Londres. 301 pp.
Jory D. E. and E. S. Iversen. (1983). Queen conch
predators: not a roadblock to mariculture.
Proceedings of the Gulf and Caribbean Fisheries
Institute, 35: 61– 75.
Keen M.A. (1971). Seashells of Tropical West America.
Stanford University, California. 1064 pp.
Lagos B., A. L. Hernández, S., R. H. Gómez and P. V.
Daza. (1996). Algunos aspectos bioecológicos y
reproductivos del caracol pala, Strombus gigas
Linnaeus, 1758 en el archipiélago de San Andres,
Providencia y Santa Catalina (Caribe
colombiano). Boletín Científico INPA, 4: 22.
Martín-Mora E., F. C. James and A. W. Stoner. (1995).
Developmental plasticity in the shell of the queen
conch Strombus gigas. Ecology, 76: 981–994.
Randall J. E. (1964). Contributions to the biology of the
queen conch, Strombus gigas. Bulletin of marine
science of the Gulf and Caribbean, 14: 246–295.
Ray-Culp M. (2002). The Conch News website:
Welcome to Conch News. En:
http://oakhammockbooks.com/conchnews Oak
Hammock Publishing.
Ray-Culp M., M. Davis and A. W. Stoner. (1997). The
micropredators of newly settled queen conch
(Strombus
gigas Linnaeus). Journal of Shellfish Research,
16: 423– 428.
Stoner A. W. and K. C. Schwarte. (1994). Queen conch,
Strombus gigas, reproductive stocks in the
central Bahamas: distribution and probable
sources. Fishery bulletin, 92:171-179.
Stoner A. W. and J. M. Waite. (1990). Distribution and
behavior of Queen Conch Strombus gigas
relative to seagrass standing crop. Fishery
bulletin, 88: 573–585.
Vega, A. and F. Pérez (2003). crecimiento del cambute,
Strombus galeatus (Gastropoda: Strombidae)
en el Parque Nacional Coiba, pacífico
panameño. Tecnociencia, 5(1): 77-89.
Villalobos, C. 1986. Biología, producción y
aprovechamiento del cambute Strombus
galeatus, en Bahía Ballena, Puntarenas, Costa
Rica. Propuesta de investigativa sometida al
Consejo Nacional de Ciencia y Tecnología, San
José, Costa Rica.
Weil, E. and R. Laughlin. (1984). Biology, population
dynamics, and reproduction of the queen
conch Strombus gigas Linne in the Archipelago
de Los Roques National Park. Journal of
Shellfish Research, 4:45-62